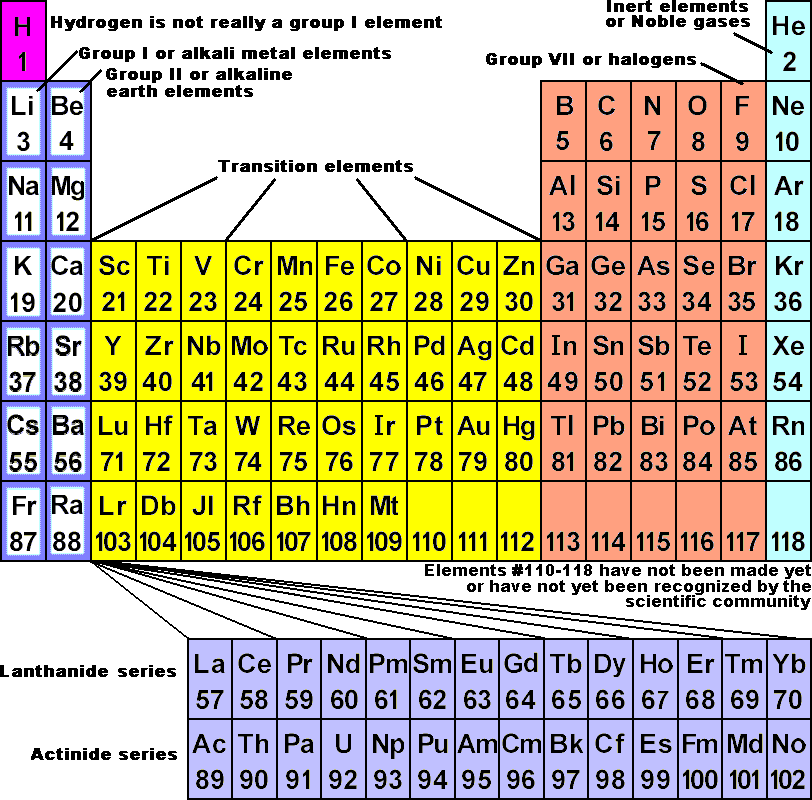
Now, the table is called the periodic table because the elements with similar properties occur at regular intervals.Ĭal: Ah, OK. Mrs Roberts: Well, elements that have similar properties to each other are put into vertical columns called groups. The way we arrange them is in the order of increasing atomic number.Ĭal: And what about these vertical columns? I mean, what does all of that mean? It has changed a lot over the years as we've learned more and more about atoms. Mrs Roberts: Well, the modern periodic table shows us all of the elements arranged in rows, which are called periods. So how has the periodic table been put together? OK, well I might have been able to find that, but I'm still a little bit lost on what all of this actually means. See if you can see it in the periodic table.Ĭal: OK. Mrs Roberts: So, the chemical symbol is Al. And what is the chemical symbol for aluminium? Now, each element has a chemical symbol made of one or two letters.Ĭal: Right, OK. Mrs Roberts: So, here I have an empty pop can. The periodic table as a list of elements arranged so as to demonstrate trends in their physical and chemical properties.Cal: Let's talk about some of the elements on the periodic table, shall we?.Describe and model the structure of the atom in terms of the nucleus, protons, neutrons and electrons comparing mass and charge of protons neutrond and electrons. Use the Periodic Table to predict the ratio of atoms in compounds of two elements. Unit 1: Structures, Trends, Chemical Reactions, Quantitative Chemistry and Analysis The remaining six elements from the periodic table which haven’t been illustrated (yet) are Ununtrium, Flerovium, Ununpentium, Livermorium, Ununseptium, Ununonctium.Unit C1: Structures, Trends, Chemical Reactions, Quantitative Chemistry and Analysis.(g) elements being arranged in order of increasing atomic number and in groups and periods in the modern Periodic Table, with elements having similar properties appearing in the same groups.2.2 ATOMIC STRUCTURE AND THE PERIODIC TABLE.(h) elements being arranged in order of increasing atomic number and in groups and periods in the modern Periodic Table, with elements having similar properties appearing in the same groups. 
1.2 ATOMIC STRUCTURE AND THE PERIODIC TABLE.Unit 1: CHEMICAL SUBSTANCES, REACTIONS and ESSENTIAL RESOURCES.(a) elements being arranged according to atomic number in the Periodic Table.Unit 1: THE LANGUAGE OF CHEMISTRY, STRUCTURE OF MATTER AND SIMPLE REACTIONS.The Periodic Table can be used to determine whether an element is a metal or non-metal.Atomic structure and bonding related to properties of materials.Elements are arranged in the periodic table in order of increasing atomic number.RSC Yusuf Hamied Inspirational Science Programme. 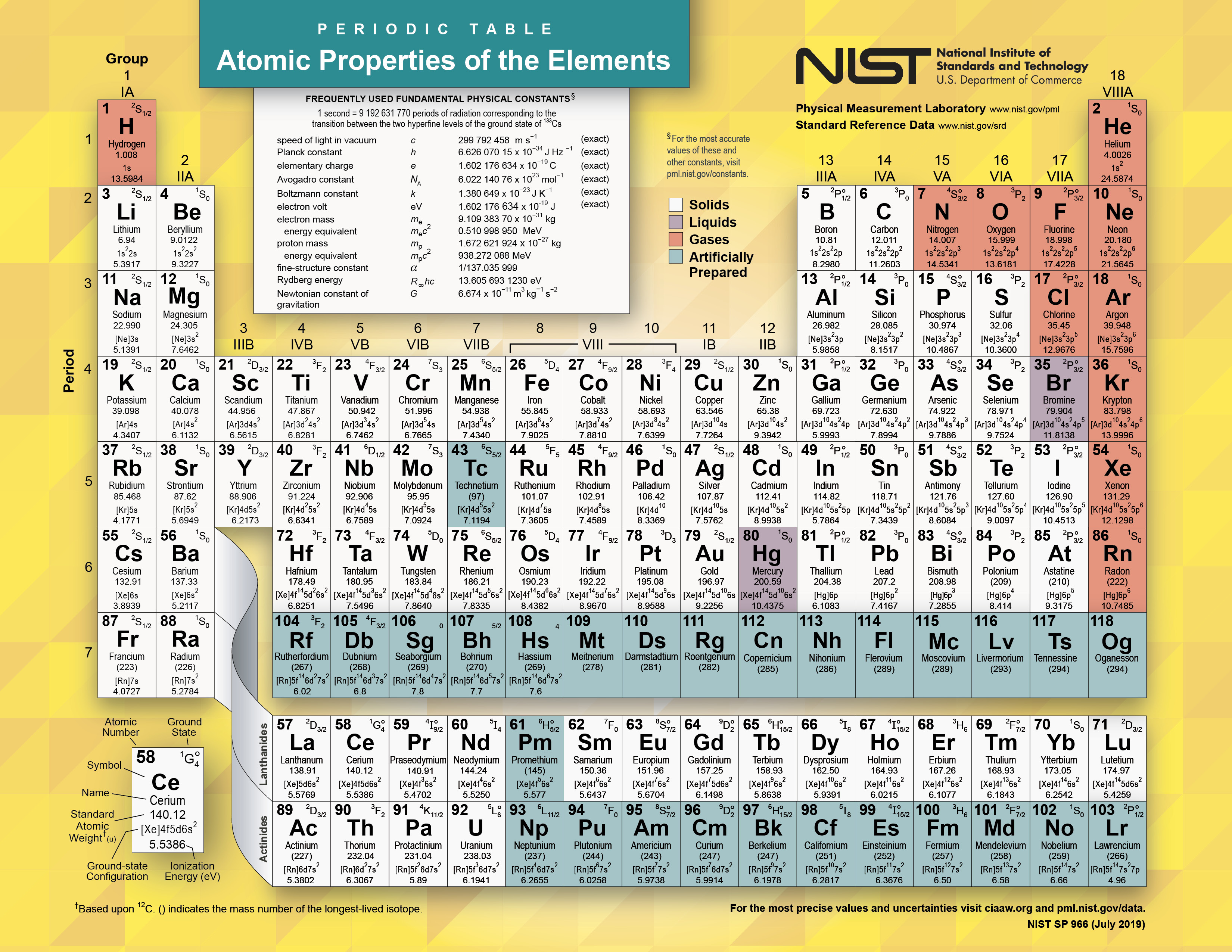
The elements are grouped based on the structure of their atomshow many protons they have and their electron configuration.
0 Comments
Leave a Reply. |



 RSS Feed
RSS Feed
